Impella Cardiac Pump Lawsuit
Were You Injured by an Impella Cardiac Pump?
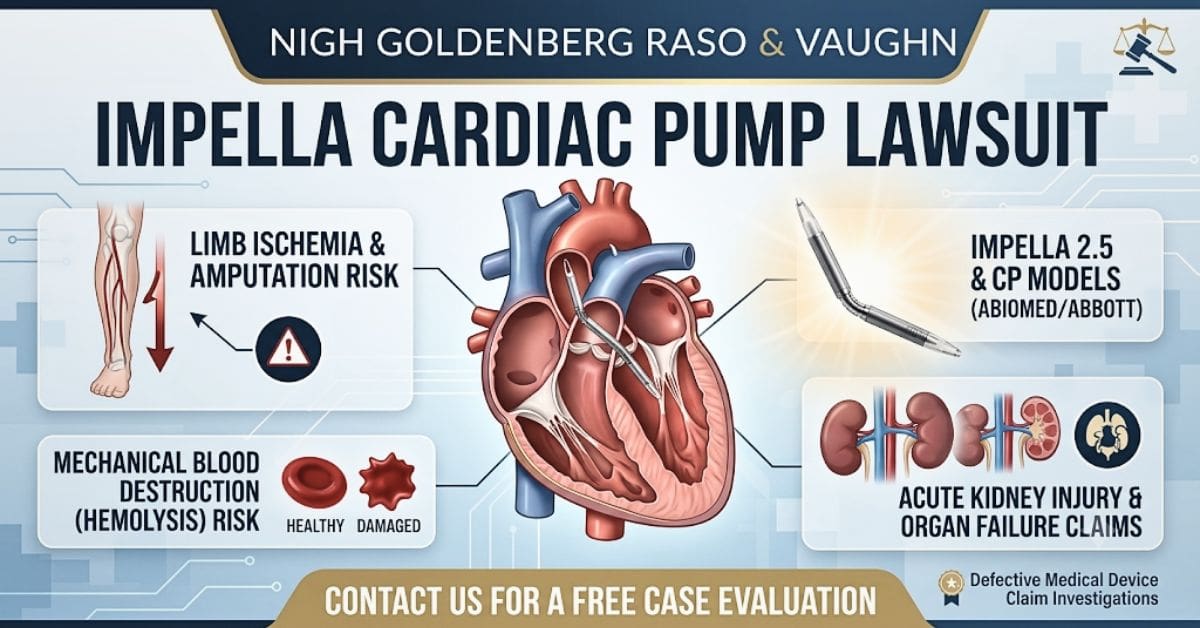
The Impella Cardiac Pump (including the Impella 2.5 and Impella CP) is a percutaneous ventricular assist device marketed to mechanically unload the heart during cardiogenic shock or high-risk cardiac procedures. For many patients, however, these devices have caused devastating complications—including severe limb ischemia leading to amputations, and mechanical blood destruction leading to organ failure.
Nigh Goldenberg Raso & Vaughn (NGRV) is actively investigating and pursuing Impella lawsuit claims on behalf of individuals harmed by these devices, which were manufactured by Abiomed (now acquired by Abbott).
If you or a loved one experienced severe complications or required an amputation after undergoing a procedure involving an Impella heart pump, you may be entitled to significant financial compensation.
Breaking Update: May 2026
- Early Mover Position: Currently, there is no consolidated Multidistrict Litigation (MDL) for Impella claims.
- Ongoing Data: Abbott’s own post-market data from the RECOVER-IV trial continues to generate alarming documentation regarding adverse events.
What Is the Impella Cardiac Pump?
The Impella is a tiny, temporary ventricular assist device (pump) inserted through the femoral artery in the leg and threaded up to sit across the aortic valve. It is designed to pump blood for the heart in patients suffering from cardiogenic shock. Abiomed originally built the device before being acquired by Abbott in 2022 for approximately $16 billion.
While promoted as a life-saving tool, mounting medical evidence indicates that these devices carry a high risk of catastrophic complications. Furthermore, multiple peer-reviewed papers have found that the Impella provides no survival benefit compared to standard-of-care intra-aortic balloon pumps, legitimately challenging the device’s harm-to-benefit ratio.
Why Are Lawsuits Being Filed?
Lawsuits allege that the manufacturer failed to adequately warn patients and doctors about severe risks. Key evidence driving this litigation includes:
- High Complication Rates in Peer-Reviewed Studies: Published clinical studies in major medical journals like JAMA and Circulation have found serious complication rates of 15% to 30% across Impella procedures.
- FDA Warnings: The FDA has issued multiple safety communications specifically warning healthcare providers about the risks of inadequate hemolysis monitoring and limb ischemia associated with Impella pumps, establishing direct notice to the manufacturer of these dangers.
Serious Injuries & Complications
Our firm is investigating claims involving severe, life-altering injuries caused by the Impella 2.5 and Impella CP devices. The two primary injuries driving the litigation theory are:
1. Limb Ischemia & Amputations
Because the Impella device occupies the femoral artery, it can severely restrict or completely cut off blood flow to the patient’s leg. This acute lower limb ischemia can cause permanent tissue death, frequently resulting in the need for partial or full leg amputations.
2. Hemolysis & Organ Failure
The fast-spinning impeller mechanism of the device can mechanically destroy red blood cells as it pumps – a condition known as hemolysis. This severe blood damage frequently leads to:
- Acute Kidney Injury (AKI)
- Severe anemia
- Multi-organ failure
Do I Qualify for an Impella Lawsuit?
You may be eligible to file a claim if:
- Implantation: You or a loved one had an Impella Cardiac Pump (specifically an Impella 2.5 or Impella CP) inserted during a cardiac procedure.
- Complications: You experienced severe side effects, specifically limb ischemia resulting in amputation, severe hemolysis resulting in kidney failure/organ damage, or wrongful death.
Potential Compensation in an Impella Lawsuit
Filing a claim is about securing the resources needed for lifelong care after a devastating medical injury. We fight for awards that account for both current and future care:
- Economic Damages (Tangible Losses)
- Medical Expenses: Coverage for past hospitalizations, revision surgeries, dialysis, amputee rehabilitation, and future life care costs.
- Lost Wages: Compensation for missed work or diminished earning capacity due to permanent disability.
- Non-Economic Damages (Quality of Life)
- Pain and Suffering: For the severe physical agony and emotional trauma of undergoing an amputation or suffering internal organ failure.
- Loss of Enjoyment of Life: For the daily activities, hobbies, and independence lost due to your injuries.
- Wrongful Death: Financial support and compensation for families who lost a loved one due to a fatal Impella complication.
How NGRV Can Help
Nigh Goldenberg Raso & Vaughn is a nationally recognized mass tort law firm uniquely positioned to handle complex Impella Cardiac Pump cases. Our team includes Attorney Brett, whose extensive cardiac nursing background provides a genuine advantage in evaluating complex cardiac case facts, interpreting medical records, and developing critical relationships with top medical experts.
When you contact NGRV:
- Your case will be reviewed for free.
- There are no upfront costs—you pay nothing unless we win.
- Our team handles every step of the legal process.
Get a Free, Confidential Case Review
The attorneys at Nigh Goldenberg Raso & Vaughn have the resources, experience, and grit to stand up to billion-dollar medical device corporations. Don’t wait—time limits apply to filing your claim.